Part Two of a two-part series on the important role of accelerometry in aging science. The Accelerometry Resource Core (ARC) at COAH has been created to help wearable data become one of the pillars of modern medicine and epidemiology by increasing its accessibility to a wide range of health researchers. To read Part One of this series, click here. To learn more about the ARC, visit accelerometry.org or reach out to Dr. Urbanek directly: jurbane2@jhu.edu or at wearables @jhu.edu.
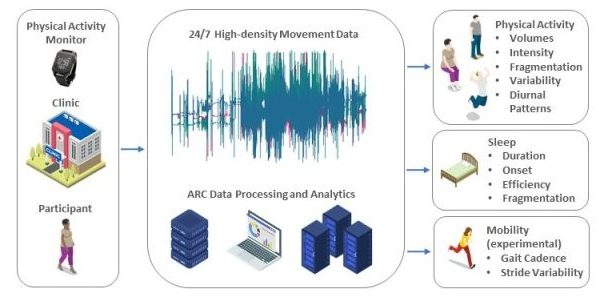
For investigators seeking to add wearable accelerometers to their research, the ARC can help with study design, including selecting devices, determining duration and frequency of monitoring, contributing to IRB applications, and contributing to funding proposals. Further, the core provides training for clinical personnel and manuals of operating procedures to ensure proper initialization and distribution of the accelerometers, data management, device maintenance, and redistribution.
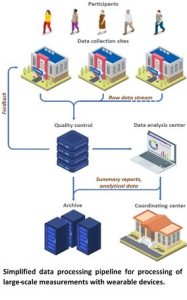
All data-related operations within ARC are handled by a proprietary, dedicated data processing platform that guarantees thorough quality control and reproducible results as soon as the next day after the collection. The platform allows for flexible, HIPAA compliant, data transfer procedures that are driven by existing file-sharing solutions at the collaborator’s institution (e.g., OneDrive, Box, SFTP, and others). After the upload, each data file is downloaded and checked for integrity, including the detection of corrupted files due to a malfunctioning device or unstable network connection. Additionally, the platform checks if the correct data has been uploaded and if the name of the file fits the study-specific convention. If any problems are identified at this stage, ARC staff immediately contacts the field center/clinic to resolve and if possible, avoid future issues of a similar nature.
| After the quality control, raw data is archived and passed on for pre-processing. As different accelerometer manufacturers use distinct, proprietary data formats, often requiring dedicated software, the ARC platform has a built-in functionality that allows communication with third-party solutions, despite the generation and type of interface. At this step, data is converted to the universal time-series format that can be then read by popular statistical analysis packages (e.g., R, Python, SAS, Stata). Next, the data is analyzed to detect if and when the accelerometer was removed by the participant during collection. Although most wearable monitors can be worn continuously, participants often choose to remove them for bathing, certain work tasks, or contact-sport activities. Proper pre-processing requires detection of such non-wear periods and, when possible, imputation of the missing data. Also, based on the detected non-wear periods, participant’s adherence to the study protocol is characterized by determining which days can be used to represent free-living movement. Clean accelerometry data is then summarized into a set of characteristics representing volumes and fragmentation of physical activity, circadian rhythms, and sleep. The summary data is next formatted to fit existing research platforms or protocols specific to each study, archived, and shared with coordinating centers. |
 |
Processing of voluminous, free-living measurements, often collected in multi-site, longitudinal settings, is challenging not only because of the complexity and sheer size of data but also due to the novelty of technology itself. At the ARC we have streamlined the measurement process and introduced measurement and analytical standards that build the foundation for a wide-scale utilization of wearable accelerometry in health research and clinical practice. To guarantee reproducibility and transparency of scientific results, ARC members created an open-source, freely available software package (ARCTOOLS for R) used in all pre-processing and analytical steps. Thanks to the creation of standards, software tools, and hardware infrastructure ARC has reduced costs for the investigators while maximizing productivity. In most cases, newly collected data is cleaned and processed into the analytical form in just a few days after the initial upload. The ARC platform is currently processing hundreds of new, high-density, multi-day observations every month in the ongoing projects of our collaborators.
After successful data collection, ARC faculty and staff offer help with statistical analyses, including a selection of physical activity characteristics that best address the aims of the study, proper adjustment for potential confounders, statistical modeling, and interpretation of the obtained results. These services are complemented with assistance in manuscript preparation, including outlining the measurement protocols and analytical approach, and interpreting the results.

An example of a Johns Hopkins Accelerometry Resource Core (ARC) partnership: Together with the ENGAGE research lab, which focuses on promoting and improving ambulatory measurement in free-living settings with wearable devices in clinical and research settings, ARC seeks to advance the science and understanding of physical activity, mobility, sleep quality, cardiovascular health, and more through advanced analytics and computational methods.
Members of ARC provide support for the collection and analytics of physical activity, heart rate, and ambulatory ECG and blood glucose measurements with wearable devices in multiple large observational studies and clinical trials. In total, ARC has collected and processed multi-day, high-density data in over 11,000 participants. Among ARC’s collaborators are:







